Cleaning & Disinfection, Knowledge Base
Disinfection of Cleanroom Surfaces With Hydrogen Peroxide
Disinfection of Surfaces with Hydrogen Peroxide Of all the contamination control activities in a cleanroom, perhaps the most critical are disinfection procedures. That’s because human health depends on the quality and thoroughness of a surface disinfection wipedown.
Many solutions can be considered for surface disinfection, but hydrogen peroxide and sodium hypochlorite (bleach) rate special attention. This article will focus on hydrogen peroxide solutions and a companion article will cover sodium hypochlorite solutions.
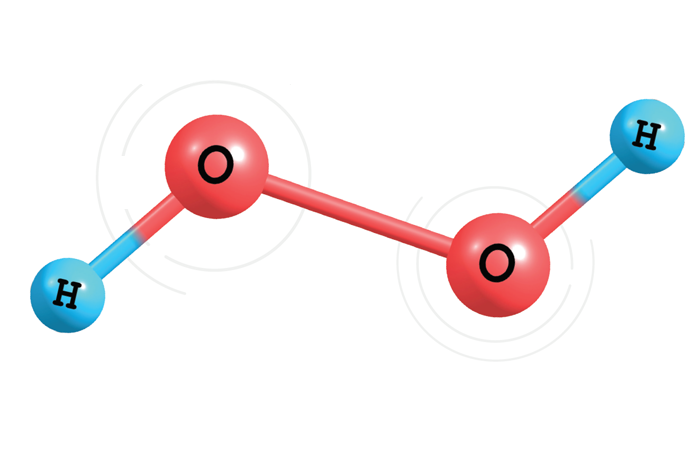
Hydrogen peroxide has been used for decades for disinfection of small wounds because of its ability to quickly destroy bacteria. Hydrogen peroxide is a simple molecule – basically two oxygen atoms bonded together, with each oxygen bonded to its own hydrogen atom – with a molecular formula of H2O2. Compared to water (H2O), hydrogen peroxide is endowed with an extra oxygen that provides it with strong oxidizing power and gives it the capability to get rid of stains and whiten surfaces. But for our purposes, that extra oxygen is responsible for destroying bacteria and other harmful organisms. The exact mechanism by which this occurs is admittedly not well understood and various theories involving hydrogen peroxide destruction of bacterial cell walls and bacterial DNA have been proposed. Note that hydrogen peroxide solutions are inherently unstable and decompose over time to oxygen gas and water. These solutions are packaged in opaque containers to prevent decomposition from external light.
Traditionally, 3 and 6% solutions of hydrogen peroxide (in water) in combination with cleanroom wipers have been utilized in cleanrooms to achieve surface sanitation and/or disinfection. Wipers can be wetted directly with a hydrogen peroxide solution in the cleanroom and the wipers applied immediately to hard surfaces for disinfection. Alternately, packaged wipers pre-wetted with hydrogen peroxide solution can be employed. This has the advantage of eliminating the dispensing of liquids in the cleanroom. However, in the pre-wetted form, the wiping cloths may accelerate the decomposition of the hydrogen peroxide within the package, effectively lowering the concentration of hydrogen peroxide applied to the surface and possibly compromising the disinfecting capability.
Since wipers wetted with hydrogen peroxide are used for disinfection of cleanrooms surfaces, special consideration should be given to wiping protocol. A major concern is that hydrogen peroxide come into contact with bare surfaces in order to kill the bacterial organisms. If there are any soils on the surface, they must be removed with detergent, then the surfaces rinsed with deionized water prior to applying hydrogen peroxide. Residual soils or detergents on the surfaces will consume hydrogen peroxide and will limit bactericidal action. If the surfaces are soil-free then direct application of hydrogen peroxide is fine.
It is essential that all surfaces be wiped thoroughly with hydrogen peroxide using the recommended linear wiping strokes. After wiping, surfaces should be allowed to air dry to maximize contact time for the liquid hydrogen peroxide to accomplish disinfection. As noted earlier, the fact that hydrogen peroxide decomposes to oxygen gas and water is a great advantage in the cleaning process, since there is no hydrogen peroxide residue to be concerned about. However, it is strongly suggested that a second wipedown of the treated surfaces be done in order to remove the endotoxins (dead bacterial cells) caused by the hydrogen peroxide. The second wipedown can either be accomplished with wipers wetted with 70% IPA, or with wipers wetted with hydrogen peroxide.



















HAVE AN IDEA FOR CONTENT?
We are always looking for ideas and topics to write about.
Contact Us